
Writing lewis structures with the octet rule. If there are too few pairs to give each atom eight electrons, change the single bonds between two atoms to either double or triple bonds by moving the unbonded pairs of electrons next to a bonding pair. List the known quantities and plan the problem. The structure on the right is the lewis electron structure, or lewis structure, for h 2 o.
Lewis dot structure sf2 full#
Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. Lewis structures show each atom and its position in the structure of the molecule using its chemical symbol. Excess electrons that form lone pairs are represented as pairs of dots, and are. For the scl2 lewis structure use the periodic table to find the total number of valence electrons for the scl2 molecule. All of the geometries listed below are examples of the five basic geometries for. The lewis structure is used to represent the covalent bonding of a molecule or ion. from 4.bp. And now that we know the total valence electrons of sf2, we will start making the lewis dot structure for this molecule. Writing lewis structures with the octet rule.Įxceptions to The Octet Rule and Variable Valence - Read. And now that we know the total valence electrons of sf2, we will start making the lewis dot structure for this molecule. This gene encodes an integral outer mitochondrial membrane protein that blocks the apoptotic death of some cells such as. * during the formation of bcl3 molecule, the boron atom contributes 3 of its valence electrons to form three bond pairs with chlorine atoms. Compare the molecular polarity of the three structures. For very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms.
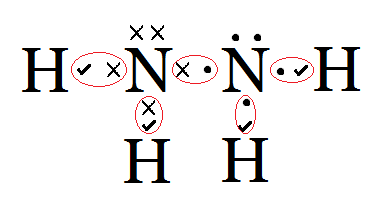
Do the same exercise for structure #2 and you find that the negative charge is on nitrogen. Draw the lewis electron dot structure for water. There is an easy way, and a formal way to draw the lewis structure of co2, carbon dioxide With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Since chlorine needs to share just one extra substances with similar chemical formulas, such as cabr2, are ionic and so their lewis structures are drawn very differently. Covalent bonds are stronger than the electrostatic interactions of ionic bonds.

If the lewis electron dot structure were drawn with a single bond between the carbon atoms and lone pairs were then added until all atoms satisfied the octet rule, it would look like this Once we know how many valence electrons there are in scl2 we can distribute them around the central atom with the goal of filling the. Chapter 10 chemical bonding and molecular structure multiple. There is an easy way, and a formal way to draw the lewis structure of co2, carbon dioxide On all unbonded sides are a single pair of dots.
Lewis dot structure sf2 how to#
You cannot really calculate a lewis structure, you just need to know how to draw them (which is really quite easy). The structure on the right is the lewis electron structure, or lewis structure, for h2o. The lewis structure for boron dichloride difluoride starts with a b atom in the center.ĬHEMIQUEST: CHEMICAL BONDS from 4.bp. Covalent bonds are a type of chemical bonding formed by the sharing of electrons in the valence shells of the atoms. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. We show two ways to draw the co2 lewis structure, carbon dioxide. We can draw the lewis structure of any covalent molecule by following the six steps discussed earlier.
